 |
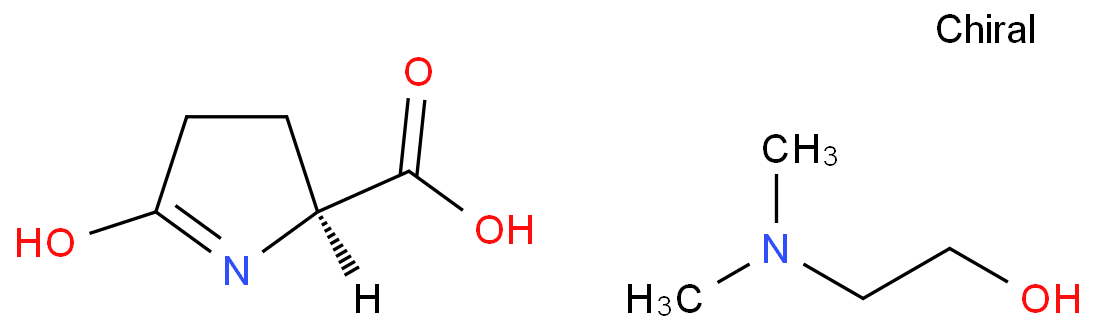 |
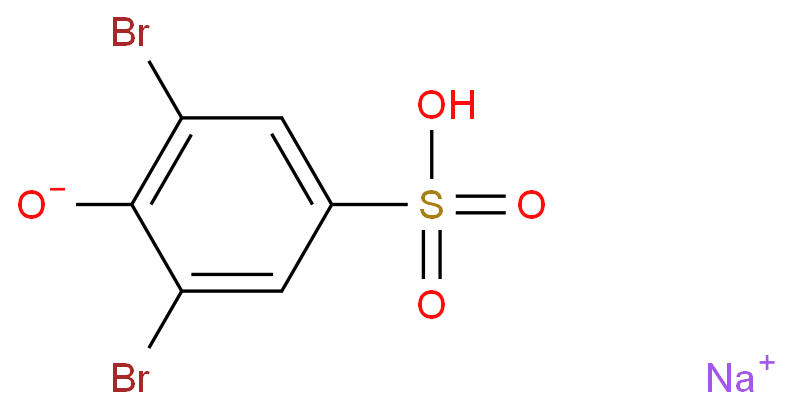 |
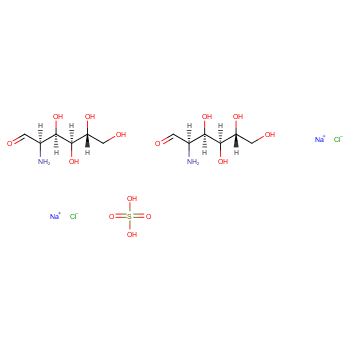 |
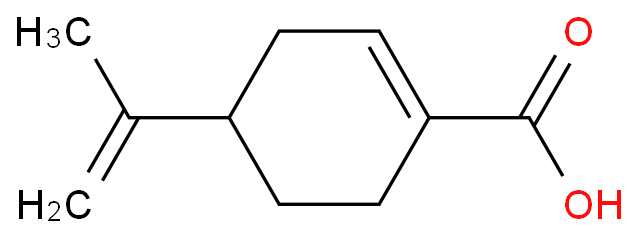 |
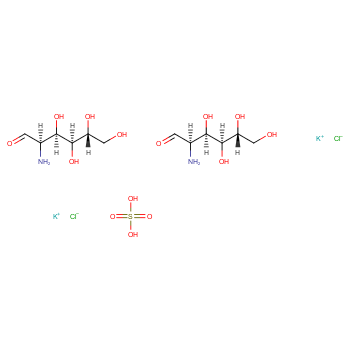 |
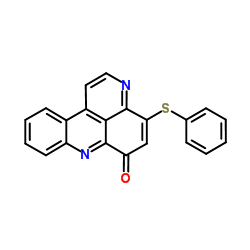 |
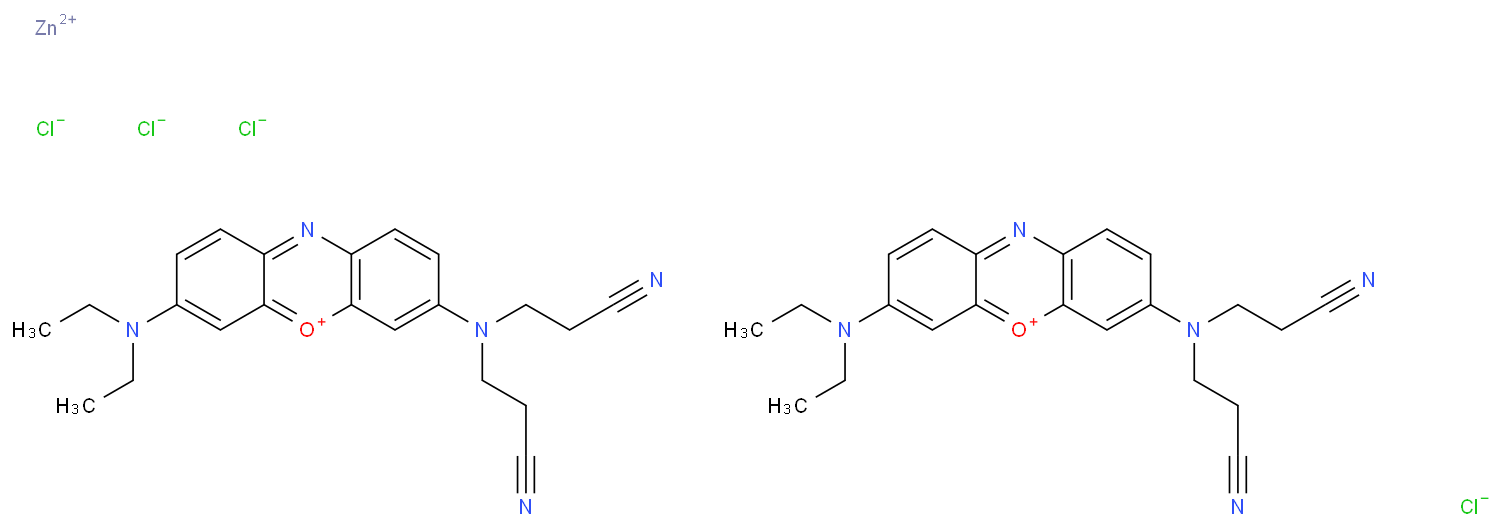 |
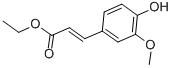 |
 |
 |
 Info marked by "
Info marked by "
The family owned business, Kraeber Co GmbH, is an independent, technology based enterprise, founded 1980. Hanseatic tradition together with courageous entrepreneurship characterize our Company.Our em...
View More >
 Kraeber & Co GmbH
Kraeber & Co GmbH